Unraveling the catalytic mechanism of TNAP through advanced molecular modelling
Published:
| Type | Research Internship |
|---|---|
| Level | Master’s student |
| Duration | 6 months |
| Start | Between September 2026 and March 2027 |
| Salary | ~ 600€/month |
| Location | ISA, Lyon, France |
| Contact | Dr Florian Blanc (florian.blanc_at_cnrs.fr), Dr. Guillaume Hoffmann (guillaume.hoffmann_at_univ-lyon1.fr), Prof. Christophe Morell (christophe.morell_at_univ-lyon1.fr) |
Download this internship offer here. Interested candidates are invited to submit a CV and a 1-page cover letter outlining their motivation, relevant skills, and research interests, to Florian Blanc, Guillaume Hoffmann and Christophe Morell at the e-mail addresses given above.
Context
Tissue-Non-specific Alkaline Phosphatase (TNAP) is an ubiquitous enzyme that cleaves phosphate groups from a wide range of substrates. TNAP is involved in biochemical processes such as bone mineralization, neurotransmitter signalling, and has emerging roles in thermogenesis and cancers. This makes it a promising target for innovative therapies in cancer and obesity. Key questions remain unanswered about TNAP’s catalytic mechanism:
- How can the TNAP active site accommodate very diverse substrates?
- How do small pH changes result in large changes in enzymatic activity?
- What is the atomic-level mechanism of phosphate cleavage?
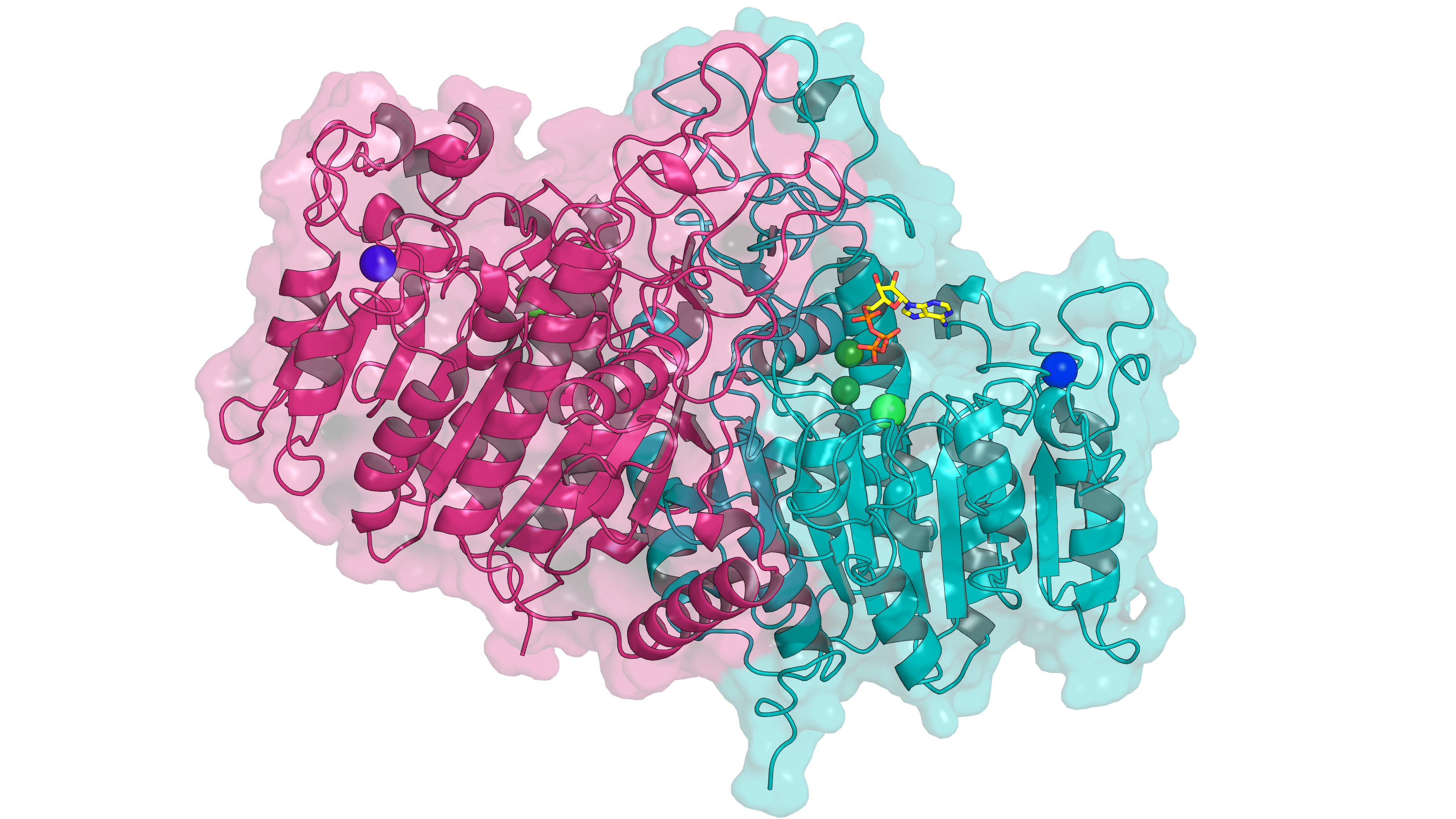
Project
Our experimental collaborators have recently solved high-resolution structures of TNAP bound to several substrates, putting us in an ideal position to investigate the enzymatic activity of TNAP at atomic resolution. This Master’s project will integrate classical Molecular Dynamics (MD) simulations and Density Functional Theory (DFT) to understand TNAP’s conformational dynamics, substrate binding properties, and catalytic mechanism. The intern will:
- Perform MD simulations of unpublished TNAP structures to characterize conformational dynamics and substrate stability.
- Use pKA predictors to identify key titratable residues and evaluate the impact of protonation states on substrate binding.
- Develop a minimal active site model and apply DFT to compute reaction pathways and energy profiles for phosphate cleavage.
We offer:
The intern will acquire complementary skills in computational chemistry and biophysics to tackle this challenging biochemical problem:
- Advanced molecular modelling (MD, DFT)
- Protein structural biology and biophysics
- Data analysis and scientific programming
- Collaboration between computational and experimental experts
We seek:
A motivated Master’s student with a taste for interdisciplinary research and a strong background in theoretical chemistry, computational biophysics, or related fields. Experience with MD simulations and/or DFT, and programming skills (Python, Linux) are expected. Familiarity with protein structural biology is desirable but not mandatory.
